Besides the binding energy BE, another important parameter for a nuclide is the binding energy per nucleon BE/A. As the name suggests, BE/A is simply the BE of a nuclide divided by the number of nucleons A in that nuclide. While BE represents the energy required to separate all the nucleons, BE/A roughly represents the energy to remove one nucleon from the nucleus. Since a nuclear transmutation occurs as long as one nucleon is separated from the nucleus, BE/A is a better indicator of the stability of a nuclide than BE. (Kind of like the GDP per capital is a better indicator of a country’s affluence level than the GDP).
| Nuclide | A | BE (MeV) | BE/A (MeV) |
| Deuterium-2 | 2 | 2.23 | 1.12 |
| Helium-4 | 4 | 28.29 | 7.07 |
| Lithium-7 | 7 | 40.15 | 5.74 |
| Beryllium-9 | 9 | 58.13 | 6.46 |
| Iron-56 | 56 | 492.24 | 8.79 |
| Lead-206 | 206 | 1622.27 | 7.88 |
| Polonium-210 | 210 | 1645.16 | 7.83 |
| Uranium-235 | 235 | 1783.80 | 7.59 |
| Uranium-238 | 238 | 1801.63 | 7.57 |
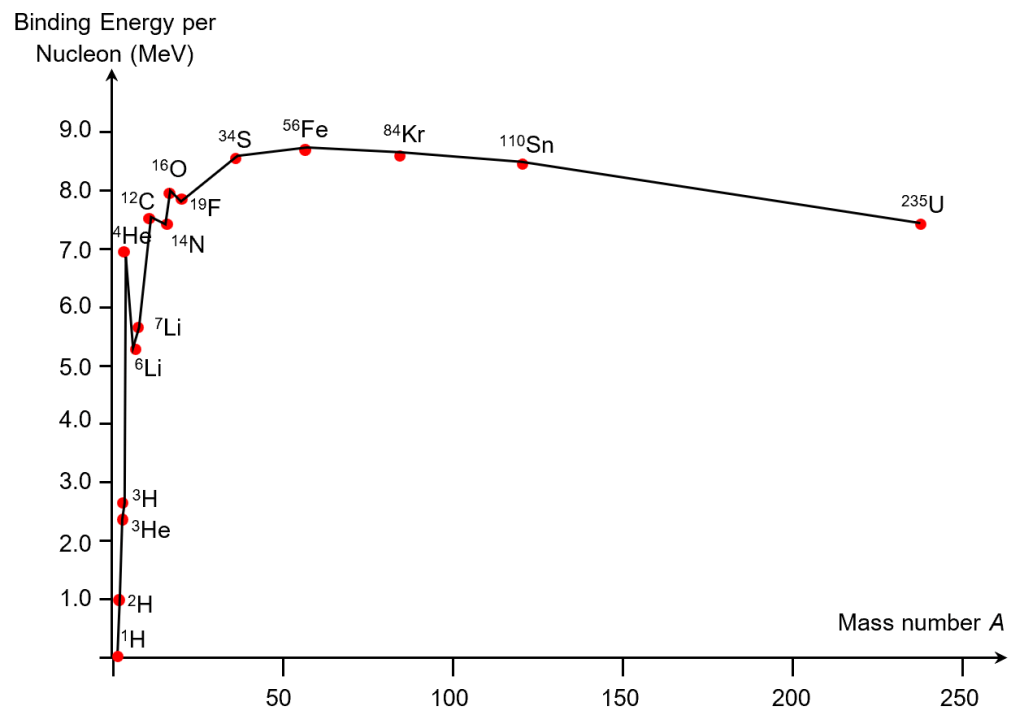
An interesting trend presents itself when BE/A is plotted against A.
Note that:
- The general trend is that BE/A increases with A for small nuclei (A<56) but decreases with A for large nuclei (A>56).[1]
- Iron-56, with the highest BE/A of 8.8 MeV, is recognized as the most strongly bound and most stable nuclide in the world.
- When small nuclei undergo fusion, the product nuclei tend to have higher BE/A than the reactant nuclei. A good example is the deuterium-tritium fusion.
- Conversely, when large nuclei undergo fission, the product nuclei tend to have higher BE/A than the reactant nuclei. A good example is the uranium fission.
- Since the number of nucleons remain the same in a nuclear reaction, a higher BE/A also implies higher total BE. This explains why energy is released by the fusion of small nuclei (e.g. hydrogen), and the fission of large nuclei (e.g. uranium).
–
[1] If you’re interested to know the reason behind this trend, and why He-4 and O-16 are outliers, see Appendix B:Nuclear Force and Appendix C:Magic Numbers.